Abstract
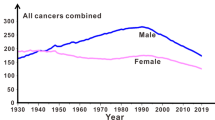
The presence of disseminated tumour cells (DTCs) in bone marrow is predictive of poor metastasis-free survival of patients with breast cancer with localized disease. DTCs persist in distant tissues despite systemic administration of adjuvant chemotherapy. Many assume that this is because the majority of DTCs are quiescent. Here, we challenge this notion and provide evidence that the microenvironment of DTCs protects them from chemotherapy, independent of cell cycle status. We show that chemoresistant DTCs occupy the perivascular niche (PVN) of distant tissues, where they are protected from therapy by vascular endothelium. Inhibiting integrin-mediated interactions between DTCs and the PVN, driven partly by endothelial-derived von Willebrand factor and vascular cell adhesion molecule 1, sensitizes DTCs to chemotherapy. Importantly, chemosensitization is achieved without inducing DTC proliferation or exacerbating chemotherapy-associated toxicities, and ultimately results in prevention of bone metastasis. This suggests that prefacing adjuvant therapy with integrin inhibitors is a viable clinical strategy to eradicate DTCs and prevent metastasis.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
Code availability
Codes used in this study (for example, ImageJ macros for image analysis) are freely available from the corresponding author upon reasonable request.
Data availability
RNA-seq data that support the findings of this study have been deposited in the GEO under accession code GSE119153. MS data have been deposited in the ProteomeXchange Consortium via the PRIDE partner repository with the dataset identifier PXD012002. Source data for Figs. 1c,f–h, 2b,c,e,g–i,k,m, 3a,b,e,h, 4g,i, 5c,e–g,i–m, 6b,e,h,j,l and 7b,f,h,j, and Supplementary Figs. 1c–e, 3a–l, 4b,c, 5b,d,e,g,h,j and 6a–d have been provided as Supplementary Table 1. All other data supporting the findings of this study are available from the corresponding author upon reasonable request.
References
Karrison, T. G., Ferguson, D. J. & Meier, P. Dormancy of mammary carcinoma after mastectomy. J. Natl Cancer Inst. 91, 80–85 (1999).
Pan, H. et al. 20-Year risks of breast-cancer recurrence after stopping endocrine therapy at 5 years. N. Engl. J. Med. 377, 1836–1846 (2017).
Braun, S. et al. Lack of effect of adjuvant chemotherapy on the elimination of single dormant tumor cells in bone marrow of high-risk breast cancer patients. J. Clin. Oncol. 18, 80–86 (2000).
Ghajar, C. M. Metastasis prevention by targeting the dormant niche. Nat. Rev. Cancer 15, 238–247 (2015).
Janni, W. et al. Persistence of disseminated tumor cells in the bone marrow of breast cancer patients predicts increased risk for relapse—a European pooled analysis. Clin. Cancer Res. 17, 2967–2976 (2011).
Sosa, M. S., Bragado, P. & Aguirre-Ghiso, J. A. Mechanisms of disseminated cancer cell dormancy: an awakening field. Nat. Rev. Cancer 14, 611–622 (2014).
Klein, C. A. Parallel progression of primary tumours and metastases. Nat. Rev. Cancer 9, 302–312 (2009).
Naume, B. et al. Clinical outcome with correlation to disseminated tumor cell (DTC) status after DTC-guided secondary adjuvant treatment with docetaxel in early breast cancer. J. Clin. Oncol. 32, 3848–3857 (2014).
Gradishar, W. J. et al. NCCN guidelines insights: breast vancer, version 1.2017. J. Natl Compr. Canc. Netw. 15, 433–451 (2017).
Jatoi, I. et al. Time-varying effects of breast cancer adjuvant systemic therapy. J. Natl Cancer Inst. 108, djv304 (2016).
Thurm, H. et al. Rare expression of epithelial cell adhesion molecule on residual micrometastatic breast cancer cells after adjuvant chemotherapy. Clin. Cancer Res. 9, 2598–2604 (2003).
Mansi, J. L. et al. Bone marrow micrometastases in primary breast cancer: prognostic significance after 6 years’ follow-up. Eur. J. Cancer 27, 1552–1555 (1991).
Braun, S. et al. A pooled analysis of bone marrow micrometastasis in breast cancer. N. Engl. J. Med. 353, 793–802 (2005).
Naumov, G. N. et al. Ineffectiveness of doxorubicin treatment on solitary dormant mammary carcinoma cells or late-developing metastases. Breast Cancer Res. Treat. 82, 199–206 (2003).
Pantel, K. et al. Differential expression of proliferation-associated molecules in individual micrometastatic carcinoma cells. J. Natl Cancer Inst. 85, 1419–1424 (1993).
Boyerinas, B. et al. Adhesion to osteopontin in the bone marrow niche regulates lymphoblastic leukemia cell dormancy. Blood 121, 4821–4831 (2013).
Cao, Z. et al. Angiocrine factors deployed by tumor vascular niche induce B cell lymphoma invasiveness and chemoresistance. Cancer Cell 25, 350–365 (2014).
Cao, Z. et al. Molecular checkpoint decisions made by subverted vascular niche transform indolent tumor cells into chemoresistant cancer stem cells. Cancer Cell 31, 110–126 (2017).
Damiano, J. S., Cress, A. E., Hazlehurst, L. A., Shtil, A. A. & Dalton, W. S. Cell adhesion mediated drug resistance (CAM-DR): role of integrins and resistance to apoptosis in human myeloma cell lines. Blood 93, 1658–1667 (1999).
Sun, Y. et al. Treatment-induced damage to the tumor microenvironment promotes prostate cancer therapy resistance through WNT16B. Nat. Med. 18, 1359–1368 (2012).
Weaver, V. M. et al. Beta4 integrin-dependent formation of polarized three-dimensional architecture confers resistance to apoptosis in normal and malignant mammary epithelium. Cancer Cell 2, 205–216 (2002).
Hambardzumyan, D. et al. PI3K pathway regulates survival of cancer stem cells residing in the perivascular niche following radiation in medulloblastoma in vivo. Genes Dev. 22, 436–448 (2008).
Ghajar, C. M. et al. The perivascular niche regulates breast tumour dormancy. Nat. Cell Biol. 15, 807–817 (2013).
Acharyya, S. et al. A CXCL1 paracrine network links cancer chemoresistance and metastasis. Cell 150, 165–178 (2012).
Trumpp, A., Essers, M. & Wilson, A. Awakening dormant haematopoietic stem cells. Nat. Rev. Immunol. 10, 201–209 (2010).
Winkler, I. G. et al. Vascular niche E-selectin regulates hematopoietic stem cell dormancy, self renewal and chemoresistance. Nat. Med. 18, 1651–1657 (2012).
Calvi, L. M. et al. Osteoblastic cells regulate the haematopoietic stem cell niche. Nature 425, 841–846 (2003).
Bruns, I. et al. Megakaryocytes regulate hematopoietic stem cell quiescence through CXCL4 secretion. Nat. Med. 20, 1315–1320 (2014).
Butler, J. M. et al. Endothelial cells are essential for the self-renewal and repopulation of Notch-dependent hematopoietic stem cells. Cell Stem Cell 6, 251–264 (2010).
Kobayashi, H. et al. Angiocrine factors from Akt-activated endothelial cells balance self-renewal and differentiation of haematopoietic stem cells. Nat. Cell Biol. 12, 1046–1056 (2010).
Ding, L., Saunders, T. L., Enikolopov, G. & Morrison, S. J. Endothelial and perivascular cells maintain haematopoietic stem cells. Nature 481, 457–462 (2012).
Kiel, M. J. et al. SLAM family receptors distinguish hematopoietic stem and progenitor cells and reveal endothelial niches for stem cells. Cell 121, 1109–1121 (2005).
Chen, J. Y. et al. Hoxb5 marks long-term haematopoietic stem cells and reveals a homogenous perivascular niche. Nature 530, 223–227 (2016).
Butler, J. M., Kobayashi, H. & Rafii, S. Instructive role of the vascular niche in promoting tumour growth and tissue repair by angiocrine factors. Nat. Rev. Cancer 10, 138–146 (2010).
Pantel, K., Braun, S., Passlick, B. & Schlimok, G. Minimal residual epithelial cancer: diagnostic approaches and prognostic relevance. Prog. Histochem. Cytochem. 30, 1–60 (1996).
Schmidt-Kittler, O. et al. From latent disseminated cells to overt metastasis: genetic analysis of systemic breast cancer progression. Proc. Natl Acad. Sci. USA 100, 7737–7742 (2003).
Gradishar, W. J. et al. Breast cancer version 3.2014. J. Natl Compr. Canc. Netw. 12, 542–590 (2014).
Xia, W. et al. Anti-tumor activity of GW572016: a dual tyrosine kinase inhibitor blocks EGF activation of EGFR/erbB2 and downstream Erk1/2 and AKT pathways. Oncogene 21, 6255–6263 (2002).
Oki, T. et al. A novel cell-cycle-indicator, mVenus-p27K-, identifies quiescent cells and visualizes G0-G1 transition. Sci. Rep. 4, 4012 (2014).
Bosch, M. & Franklin-Tong, V. E. Temporal and spatial activation of caspase-like enzymes induced by self-incompatibility in Papaver pollen. Proc. Natl Acad. Sci. USA 104, 18327–18332 (2007).
Hirata, E. et al. Intravital imaging reveals how BRAF inhibition generates drug-tolerant microenvironments with high integrin beta1/FAK signaling. Cancer Cell 27, 574–588 (2015).
Seguin, L. et al. An integrin beta(3)-KRAS-RalB complex drives tumour stemness and resistance to EGFR inhibition. Nat. Cell Biol. 16, 457–468 (2014).
Woelfle, U. et al. Bi-specific immunomagnetic enrichment of micrometastatic tumour cell clusters from bone marrow of cancer patients. J. Immunol. Methods 300, 136–145 (2005).
Kusumbe, A. P., Ramasamy, S. K., Starsichova, A. & Adams, R. H. Sample preparation for high-resolution 3D confocal imaging of mouse skeletal tissue. Nat. Protoc. 10, 1904–1914 (2015).
Lopes da Silva, M. & Cutler, D. F. von Willebrand factor multimerization and the polarity of secretory pathways in endothelial cells. Blood 128, 277–285 (2016).
Nakamura-Ishizu, A. et al. Extracellular matrix protein tenascin-C is required in the bone marrow microenvironment primed for hematopoietic regeneration. Blood 119, 5429–5437 (2012).
Weekes, C. D., Kuszynski, C. A. & Sharp, J. G. VLA-4 mediated adhesion to bone marrow stromal cells confers chemoresistance to adherent lymphoma cells. Leuk. Lymphoma 40, 631–645 (2001).
Dittel, B. N., McCarthy, J. B., Wayner, E. A. & LeBien, T. W. Regulation of human B-cell precursor adhesion to bone marrow stromal cells by cytokines that exert opposing effects on the expression of vascular cell adhesion molecule-1 (VCAM-1). Blood 81, 2272–2282 (1993).
Day, C. P. et al. "Glowing head" mice: a genetic tool enabling reliable preclinical image-based evaluation of cancers in immunocompetent allografts. PLoS ONE 9, e109956 (2014).
Holliday, D. L. & Speirs, V. Choosing the right cell line for breast cancer research. Breast Cancer Res. 13, 215 (2011).
Harrell, J. C. et al. Genomic analysis identifies unique signatures predictive of brain, lung, and liver relapse. Breast Cancer Res. Treat. 132, 523–535 (2012).
Copson, E. et al. Prospective observational study of breast cancer treatment outcomes for UK women aged 18-40 years at diagnosis: the POSH study. J. Natl Cancer Inst. 105, 978–988 (2013).
Hall, D. E. et al. The alpha 1/beta 1 and alpha 6/beta 1 integrin heterodimers mediate cell attachment to distinct sites on laminin. J. Cell Biol. 110, 2175–2184 (1990).
Cheresh, D. A. Human endothelial cells synthesize and express an Arg-Gly-Asp-directed adhesion receptor involved in attachment to fibrinogen and von Willebrand factor. Proc. Natl Acad. Sci. USA 84, 6471–6475 (1987).
Li, W., Germain, R. N. & Gerner, M. Y. Multiplex, quantitative cellular analysis in large tissue volumes with clearing-enhanced 3D microscopy (Ce3D). Proc. Natl Acad. Sci. USA 114, E7321–E7330 (2017).
Park, C. C. et al. Beta1 integrin inhibitory antibody induces apoptosis of breast cancer cells, inhibits growth, and distinguishes malignant from normal phenotype in three dimensional cultures and in vivo. Cancer Res. 66, 1526–1535 (2006).
Brooks, P. C. et al. Antiintegrin alpha v beta 3 blocks human breast cancer growth and angiogenesis in human skin. J. Clin. Invest. 96, 1815–1822 (1995).
Hawkins, E. D. et al. Regulation of asymmetric cell division and polarity by Scribble is not required for humoral immunity. Nat. Commun. 4, 1801 (2013).
Gilbert, L. A. & Hemann, M. T. DNA damage-mediated induction of a chemoresistant niche. Cell 143, 355–366 (2010).
Gomez-Sarosi, L., Sun, Y., Coleman, I., Bianchi-Frias, D. & Nelson, P. S. DNA damage induces a secretory program in the quiescent TME that fosters adverse cancer phenotypes. Mol. Cancer Res. 15, 842–851 (2017).
Miller, B. E., Miller, F. R., Wilburn, D. J. & Heppner, G. H. Analysis of tumour cell composition in tumours composed of paired mixtures of mammary tumour cell lines. Br. J. Cancer 56, 561–569 (1987).
Nombela-Arrieta, C. et al. Quantitative imaging of haematopoietic stem and progenitor cell localization and hypoxic status in the bone marrow microenvironment. Nat. Cell Biol. 15, 533–543 (2013).
Briand, P., Nielsen, K. V., Madsen, M. W. & Petersen, O. W. Trisomy 7p and malignant transformation of human breast epithelial cells following epidermal growth factor withdrawal. Cancer Res. 56, 2039–2044 (1996).
Tang, Y. et al. MT1-MMP-dependent control of skeletal stem cell commitment via a beta1-integrin/YAP/TAZ signaling axis. Dev. Cell 25, 402–416 (2013).
Kim, D. et al. TopHat2: accurate alignment of transcriptomes in the presence of insertions, deletions and gene fusions. Genome Biol. 14, R36 (2013).
Robinson, M. D., McCarthy, D. J. & Smyth, G. K. edgeR: a Bioconductor package for differential expression analysis of digital gene expression data. Bioinformatics 26, 139–140 (2010).
Subramanian, A. et al. Gene set enrichment analysis: a knowledge-based approach for interpreting genome-wide expression profiles. Proc. Natl Acad. Sci. USA 102, 15545–15550 (2005).
Szklarczyk, D. et al. The STRING database in 2017: quality-controlled protein–protein association networks, made broadly accessible. Nucleic Acids Res. 45, D362–D368 (2017).
Barrett, A. S. et al. Hydroxylamine chemical digestion for insoluble extracellular matrix characterization. J. Proteome Res. 16, 4177–4184 (2017).
Goddard, E. T. et al. Quantitative extracellular matrix proteomics to study mammary and liver tissue microenvironments. Int. J. Biochem. Cell Biol. 81, 223–232 (2016).
Acknowledgements
The authors thank the following people: G. Merlino and C.-P. Day for providing C.FVB-Tg(GNrhr-luc/EGFP)L8Mrln/LmwJ mice; A. Putnam for providing human umbilical vein endothelial cells; S. Beronja for providing murine targeted shRNA constructs; P. Paddison for providing the mVenus-p27K– vector; C. Morse for instruction on bilateral ovariectomy; S. Hingorani for in-depth discussions about preclinical trial design; and P. Lampe for instruction regarding antibody purification. They are also thankful to S. Beronja for his critical feedback on this work. This study was catalysed by start-up funds provided by the Fred Hutchinson Cancer Research Center and a grant from the Cuyamaca Foundation (to C.M.G.), and supported to its completion by an Era of Hope Award from the Department of Defense (DoD) Breast Cancer Research Program (BCRP; W841XWH-15-1-0201 to C.M.G. and K.C.H.), a grant from the Breast Cancer Research Foundation (IIDRP-17-001 to C.M.G.), a grant from the National Breast Cancer Coalition’s Artemis Project for Metastasis Prevention, a Physical Sciences Oncology Project Grant from the NIH/NCI (U54CA193461-01 to C.M.G.), and by the Comparative Medicine, Antibody Technology, Experimental Histopathology and Genomics Shared Resources of the Fred Hutchinson/University of Washington Cancer Consortium (P30 CA015704). P.S.N.’s laboratory is supported by the Prostate Cancer Foundation and NIH/NCI grants P50CA097186, U54CA224079 and R01CA165573, and by the DoD awards PC170431 and PC160662. K.C.H.’s laboratory is also supported by a grant from the NIH/NCI (R33CA183685). A.D. is supported by a fellowship from the Terri Brodeur Breast Cancer Foundation, C.A.G. is supported by a postdoctoral fellowship from the Susan G. Komen Foundation, J.D. is supported by a Postdoctoral Breakthrough award by the DoD’s BCRP, A.R.L. was supported by a Cardiovascular Pathology Training Grant from the NIH (5T32HL007213-38) and is currently supported by a fellowship from the NIH/NCI (1F31CA228424-01), and S.B.C. is supported by a Cellular and Molecular Biology Training Grant from the NIH (T32GM007270).
Author information
Authors and Affiliations
Contributions
P.C., A.D., C.A.G., J.K., R.E.S., E.T.G., J.D., E.M.S., A.R.L. and S.B.C. performed experiments and analysed data. I.M.C. and P.S.N. conducted all procedures related to RNA sequencing, and analysed and interpreted resulting data. A.B. and K.C.H. conducted all ECM-targeted MS, and analysed and interpreted the resulting data. D.A.C. provided critical reagents. D.A.C., P.S.N. and K.C.H. provided scientific insight. C.M.G. conceived the study, conducted experiments, analysed and interpreted data, and wrote the manuscript. All authors read and provided feedback on the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Integrated supplementary information
Supplementary Figure 1 Dose-dense chemotherapeutic regimens select for perivascular disseminated tumour cells in tibia.
Tibia from the experiment detailed in Fig. 1a were stained and whole-mounted. Tile scans encompassing the full area of the bone and the full Z-depth that could be imaged using this method were acquired. n = 228 DTCs were analyzed across three tibia from three AC-treated mice, and a) binned in histogram format or b) dot-plotted as individual distances to the nearest blood vessel (red), megakaryocyte (gray) or osteoblast (black). c-e) Average of these distances to the nearest c) blood vessel, d) megakaryocyte and e) osteoblast for mice that underwent treatment with vehicle (n = 286 DTCs analyzed) or dose-dense AC (n = 228). *** P < 0.0001; all P values obtained via unpaired, two-tailed t test. f-h) the percentage of DTCs on (i.e., with a plotted distance of zero) h) blood vessels, i) megakaryocytes and j) osteoblasts in tibia of mice that underwent treatment with vehicle or dose-dense AC. For c-e, centre line represents the mean, error bars the s.e.m. Statistical tests and P values are indicated. Source data are provided in Supplementary Table 1.
Supplementary Figure 2 Endothelium does not bias mesenchymal stem cells towards or away from osteo- and adipo-genic lineages in bone marrow MVNs.
a) Gene set enrichment analysis of GO “Osteoblast Differentiation” genes and b) Hallmark “Adipogenesis” genes in bone marrow MVNs and bone marrow stroma (derived from analysis of (n = 2 biologically independent samples). The enrichment statistic was computed using the Weighted Kolmogorov Smirnov test, and controlled for multiple testing by false discovery rate. Neither indicate significant enrichment in bone marrow MVNs. P-values and FDR q values listed atop figure. Follow-up staining of bone marrow stroma (MSC) and bone marrow MVNs (MSC+EC) for calcium deposits and lipid droplets via c) the von Kossa method and d) Oil Red O stain. Representative images from n = 3 independent experiments shown.
Supplementary Figure 3 Microvascular endothelium protects basal breast tumour cells from other therapies and luminal/ER+ breast tumour cells from doxorubicin.
T4-2 cells were seeded on bone marrow stroma or on bone marrow MVNs, as described in Fig. 2a, and treated with increasing doses of 5-FU or lapatinib. Dose response of T4-2 ‘DTCs’ to 5-FU on bone marrow a) stroma and b) MVNs (n = 3 independent experiments). NS denotes no significance, whereas * P = 0.020 and ** P = 0.005 by one-way ANOVA and Dunnett’s multiple comparison test, comparing the indicated dose to vehicle treatment. The percentage of TUNEL-positive cells was quantified across n = 3 independent experiments for c) stroma and d) MVNs. NS, no significance, *** P < 0.0001 by one-way ANOVA and Dunnett’s post-test compared to vehicle treatment. Dose response of T4-2 cells to lapatinib on bone marrow e) stroma and f) MVNs (n = 5 independent experiments). NS, no significance, * P = 0.038, ** P = 0.0038 by one-way ANOVA and Dunnett’s post-test, compared to vehicle treatment. g) The percentage of TUNEL-positive cells was quantified across n = 3 independent experimentsfor g) stroma and h) MVNs. NS, no significance, *** P < 0.0001 when comparing the condition indicated to vehicle by one-way ANOVA and Dunnett’s post-test. Dose response of MCF-7 breast tumour ‘DTCs’ to doxorubicin on bone marrow i) stroma and j) MVNs (n = 5 independent experiments). NS, no significance, * P = 0.0373, *** P = 0.0007 when comparing the indicated condition(s) to vehicle (0 nM) by one-way ANOVA and Dunnett’s post-test. TUNEL intensity within YFP+ tumour cells/cell clusters was quantified on bone marrow k) stroma and l) MVNs across n = 3 independent experiments. *** P < 0.0001. Indicated P values obtained by unpaired two-tailed t test. For TUNEL analyses, the number of cells (predominantly single cells and clusters of 2–4 cells) analyzed across n = 3 independent experiments is stated within each panel. For a-l, centre line represents the mean, error bars the s.e.m. Statistical tests and P values are indicated. Source data are provided in Supplementary Table 1.
Supplementary Figure 4 IGF-1 triggers quiescent breast tumour cells to re-enter the cell cycle.
Top: To assess the effect of IGF-1 on the phenotype of perivascular breast tumour cells, bone marrow MVNs were primed with 300 ng/mL IGF-1 at days 10, 12 and 15 (vs. vehicle control) a) Representative images of quiescent, microvascular-associated T4-2 cells at Day 10 (left), and after priming with vehicle or with 300 ng/mL IGF-1 at Day 17 (middle). Cultures were fixed and stained at this time point to analyze the proliferative status of tumour cells (right). Scale bars, 50 μm. White arrowhead denotes a Ki67+ tumour cell within an IGF-1 primed culture. b) Tumour cell outgrowth between day 10 and day 17 after priming with vehicle or IGF-1, normalized to vehicle control (n = 5 independent experiments). ** P = 0.0016 by unpaired, two-tailed t test. c) Percentage of Ki67+ DTCs/DTC cluster at day 17 after priming with vehicle or 300 ng/mL IGF-1. n = 31 (Vehicle) and n = 32 (Primed) DTCs were analyzed across n = 3 independent experiments. *** P = 0.0002 by unpaired, two-tailed t test. For b-c, centre line represents the mean, error bars the s.e.m. Statistical tests and P values are indicated. Source data are provided in Supplementary Table 1.
Supplementary Figure 5 Knockdown studies confirm that integrin inhibition functions specifically in the tumour cell compartment to induce chemosensitization, without affecting cell survival.
a) Representative immunoblots of integrin-β1 and the nuclear envelope proteins Lamin A/C (used as a loading control). ‘K’ depicts the Kaleidoscope molecular weight marker. Lanes 1 and 3 are lysates of independently generated T4-2 cells transduced with non-targeting shRNA (shNT). Lanes 2 and 4 are lysates of independently generated T4-2 cells transduced with shRNA targeting ITGB1 (shITGB1, clone 391001). b) Densitometry from n = 3 independent experiments, normalized to the band intensity of shNT control. *** P < 0.0001 by unpaired, two-tailed t test. shNT and shITGB1 T4-2 cells were seeded on bone marrow MVNs as depicted in Fig. 2a and analyzed 12-days later. c) Representative images of bone marrow MVNs at this time point. Scale bar, 200 μm. d) Number of clusters and e) Tumour cell outgrowth at day 12, to assess the effect of ITGB1 knockdown on tumour cell phenotype in the absence of doxorubicin (n = 5 independent experiments). ** P = 0.0039. P values obtained by unpaired, two-tailed t test. Cultures were treated at days 12 and 15 with 2500 nM doxorubicin, then fixed and stained two days later to assess DTC viability. f) Representative images of TUNEL stained shNT and shITGB1 DTCs on bone marrow MVNs. Scale bar, 20 μm. White arrowhead, TUNEL-positive tumour cell. g) Tumour cell outgrowth over the treatment period (n = 5 independent experiments). * P = 0.050 by unpaired two-tailed t test. The total percentage of h) TUNEL-positive tumour cells, i) TUNEL-positive single DTCs, and j) TUNEL-positive cells within 2–4 cell clusters was analyzed across n = 116 (shNT) and n = 124 (shITGB1) total cells/clusters across three independent experiments. *** P < 0.0001 by unpaired, two-tailed t test. For b, d-e, g-h, j centre line represents the mean, error bars the s.e.m. Statistical tests and P values are indicated. Source data are provided in Supplementary Table 1.
Supplementary Figure 6 Integrin inhibition does not exacerbate chemotherapy-induced toxicities.
Mice treated as described in Fig. 6d were a) weighed weekly. Body weights of treated mice did not differ significantly over the course of treatment or after one week, as assessed by multiple unpaired t tests and correcting for multiple comparisons using the Holm-Sidak method. Cohort size: IgG+AC: 11 animals; AIIB2+AC: 10 animals; AIIB2+LM709+AC: 9 animals. b) Bone marrow vascular density was determined from pan-laminin staining femurs of athymic nude females from the study described in Fig. 6c (Cohort 1). Data were not statistically significant, as determined by Kruskall-Wallis test and Dunn’s post-test to correct for multiple comparisons. c) Spleens from these mice were also weighed to grossly account for changes in immune cell content. Splenic weights did not differ between treatment conditions (but all were substantially lower than mice treated only with isotype control antibody), as assessed by one-way ANOVA and Dunnett’s post-test. For (b) and (c), Cohort size: IgG+AC: 4 animals; AIIB2+AC: 4 animals; AIIB2+LM709+AC: 3 animals. d) The effect of AIIB2 and/or AC therapy on immune cell content within bone marrow of uninoculated, immune-competent FVB mice was quantified by flow cytometery to determine whether AIIB2 affected specific immune cell populations. n = 3 animals were analyzed per treatment. No significant differences in viable CD45+ leukocytes were observed. *P = 0.029 and **P = 0.0096 when comparing the number of CD11b+ cells in IgG+AC and AIIB2+AC treated mice to untreated mice. **P = 0.0011 and ***P = 0.0008 when comparing the number of B220+ cells in IgG+AC and AIIB2+AC treated mice to untreated mice; *P = 0.033 when comparing the number of CD3+ cells in IgG+AC treated mice to untreated mice (comparing CD3+ cells in AIIB2+AC vs. untreated mice yielded P = 0.073). In each case, comparing IgG+AC and AIIB2+AC treatments yielded P values >0.983. All comparisons were made using one-way ANOVA and Dunnett’s post-test. For a-d, centre line represents the mean, error bars the s.e.m. Statistical tests and P values are indicated. Source data are provided in Supplementary Table 1.
Supplementary Figure 7 Unprocessed immunoblots to assess shRNA-mediated knockdown of VWF and integrin β1.
a) Full representative immunoblot of VWF and nuclear envelope proteins Lamin A/C (used as a loading control). Lane 1 is from lysate of HUVEC transduced with non-targeting (NT) shRNA. Lane 2 is from lysate of HUVEC transduced with shRNA targeting VWF (VWF, clone 643). Lane 3 is from lysate of HUVEC transduced with shRNA targeting VWF (VWF, clone 620). Results are representative of those obtained from n = 3 independently generated lysates per condition. Densitometry from these three independent experiments is shown in Fig. 5b. b) Full representative immunoblots of integrin-β1 and the nuclear envelope proteins Lamin A/C. ‘K’ depicts the Kaleidoscope molecular weight marker. Lanes 1 and 3 are independently generated lysates from T4-2 cells transduced with non-targeting shRNA (shNT). Lanes 2 and 4 are independently generated lysates from T4-2 cells transduced with shRNA targeting ITGB1 (shITGB1, clone 391001). Results are representative of those obtained for n = 3 independently generated lysates per condition. Densitometry from these three independent experiments is shown in Supplementary Figure 5b.
Supplementary information
Supplementary Information
Supplementary Figures 1–7, and legends for Supplementary Tables and Supplementary Videos.
Supplementary Table 1
Statistics source data.
Supplementary Table 2
Primary tumour, bone marrow area, and DTC measurements related to Fig. 6l.
Supplementary Table 3
Details on antibodies used for immunofluorescent staining, immunoblotting, flow cytometry and blocking integrin function.
Supplementary Table 4
Sequence information for shRNA clones.
Supplementary Video 1
Apoptosis of p27-positive and p27-negative breast tumour cells in response to doxorubicin on bone marrow stroma.
Supplementary Video 2
Apoptosis of p27-negative breast tumour cells in response to doxorubicin on bone marrow microvascular niche culture.
Supplementary Video 3
Death of a breast tumour cell cluster containing a p27-positive and a p27-negative breast tumour cell in response to doxorubicin on a bone marrow microvascular niche culture.
Rights and permissions
About this article
Cite this article
Carlson, P., Dasgupta, A., Grzelak, C.A. et al. Targeting the perivascular niche sensitizes disseminated tumour cells to chemotherapy. Nat Cell Biol 21, 238–250 (2019). https://doi.org/10.1038/s41556-018-0267-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41556-018-0267-0
This article is cited by
-
How much do we know about the metastatic process?
Clinical & Experimental Metastasis (2024)
-
CAFs vs. TECs: when blood feuds fuel cancer progression, dissemination and therapeutic resistance
Cellular Oncology (2024)
-
A cell cycle centric view of tumour dormancy
British Journal of Cancer (2023)
-
Bone serves as a transfer station for secondary dissemination of breast cancer
Bone Research (2023)
-
Bone-matrix mineralization dampens integrin-mediated mechanosignalling and metastatic progression in breast cancer
Nature Biomedical Engineering (2023)